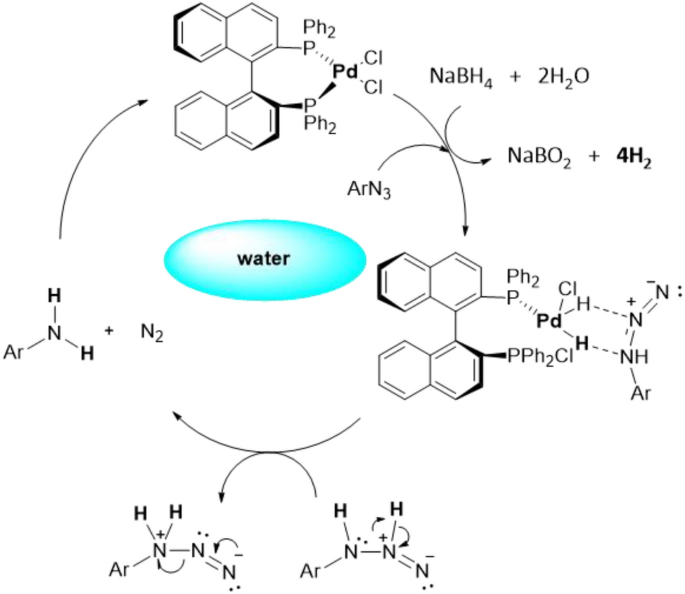
Scheme: Mechanism of palladium(II) reduction from a glycinate electrolyte. | Download Scientific Diagram
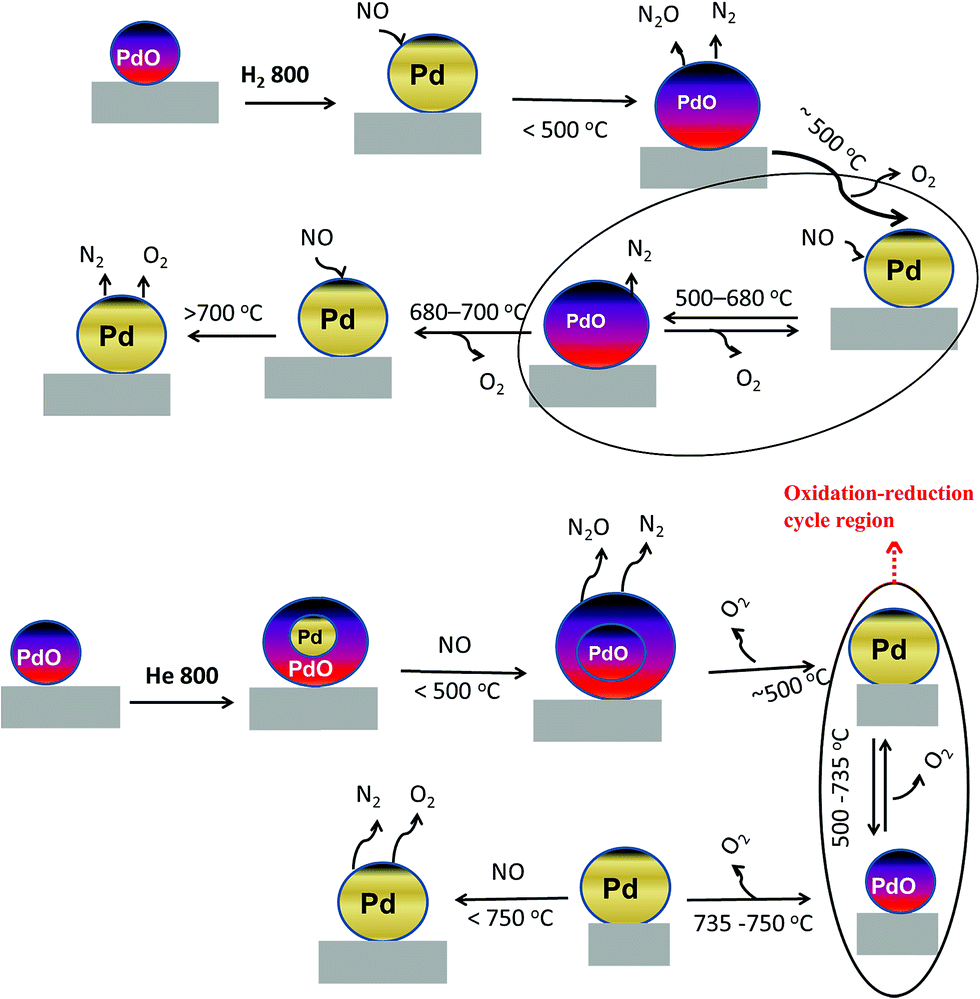
Understanding the chemical state of palladium during the direct NO decomposition – influence of pretreatment environment and reaction temperature - RSC Advances (RSC Publishing) DOI:10.1039/C7RA00836H

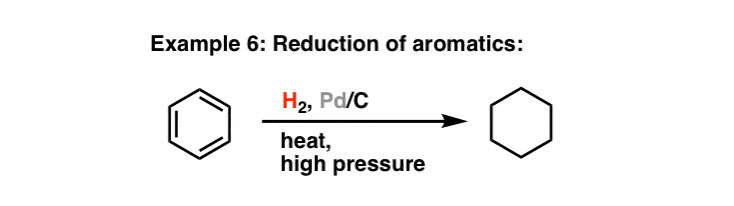
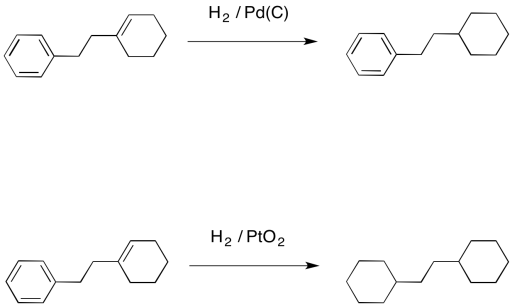
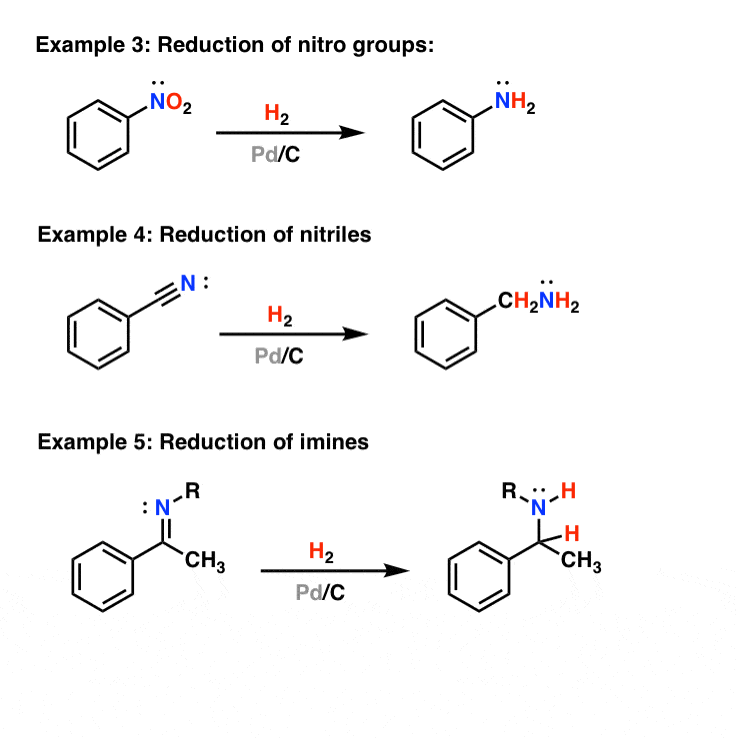
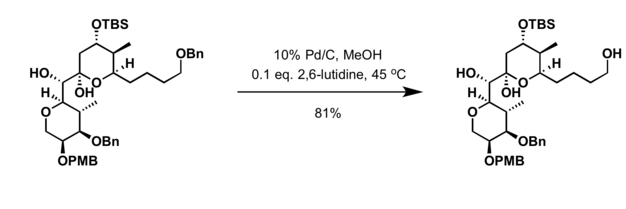
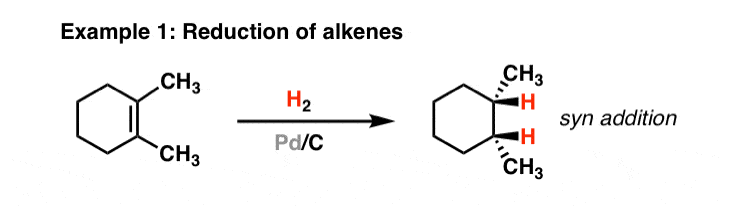
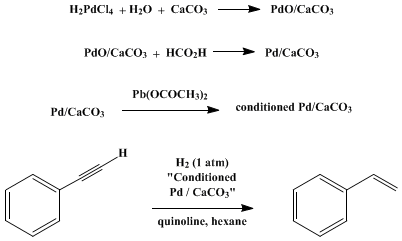
Compound A (C_9 H_{12}) absorbed 3 equivalents of H_2 on catalytic reduction over a palladium catalyst to give B (C_9 H_{18}). On ozonolysis, compound A gave, among other things, a ketone that
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img02.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

Understanding the Unusual Reduction Mechanism of Pd(II) to Pd(I): Uncovering Hidden Species and Implications in Catalytic Cross-Coupling Reactions. | Semantic Scholar

The Newman-Kwart Rearrangement of O-Aryl Thiocarbamates: Substantial Reduction in Reaction Temperatures through Palladium Catalysis
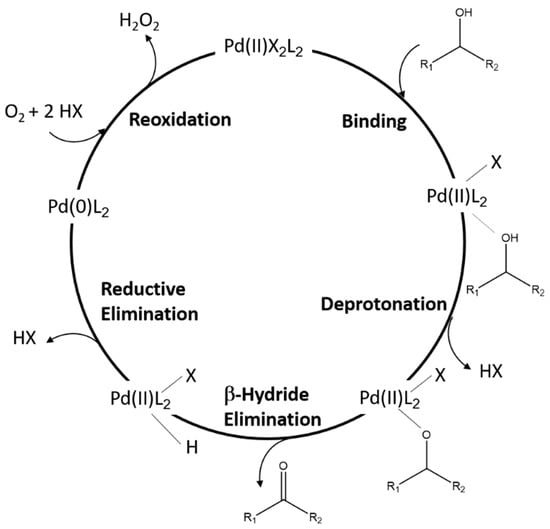
Metals | Free Full-Text | The Effect of Alcohol on Palladium Nanoparticles in i-Pd(OAc)2(TPPTS)2 for Aerobic Oxidation of Benzyl Alcohol
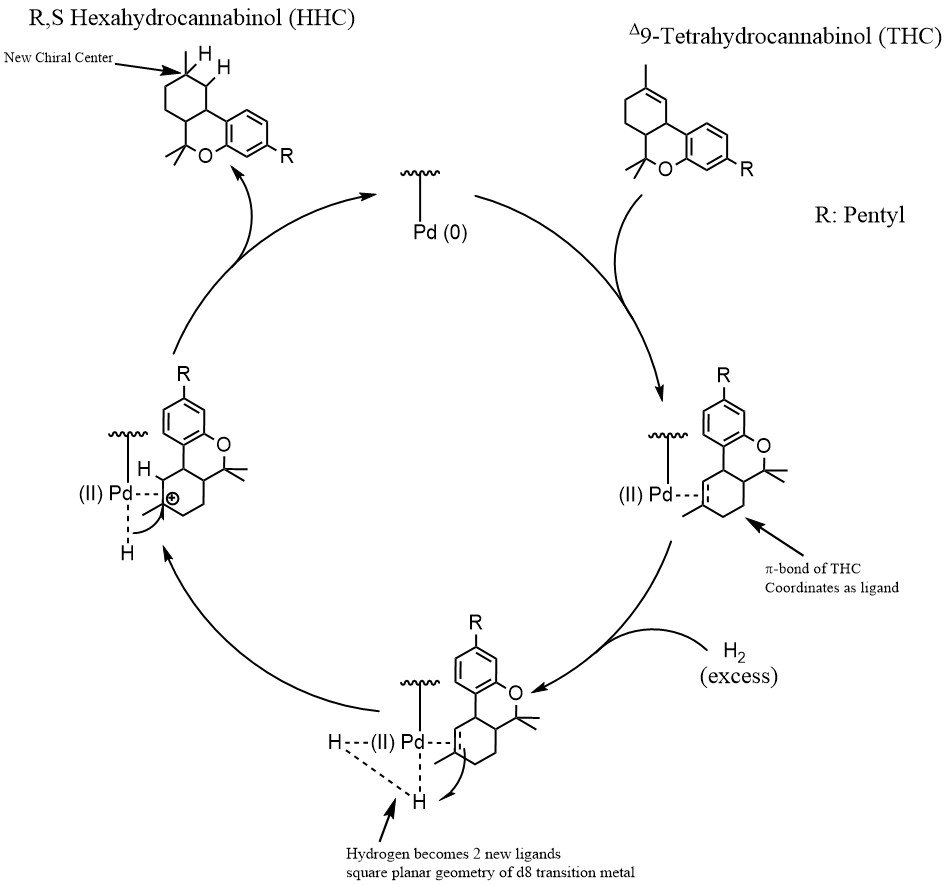
Organometallic Chemistry: Reduction, Oxidation, Metathesis & More! - Pre/Post-Processing - Future4200
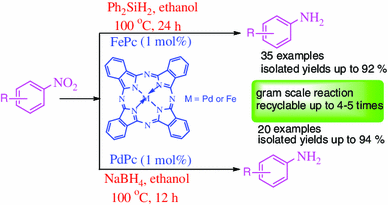
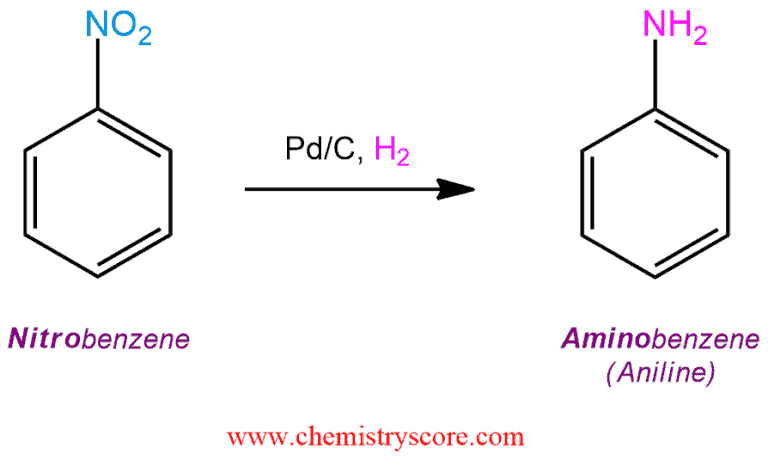
Iron and Palladium(II) Phthalocyanines as Recyclable Catalysts for Reduction of Nitroarenes | SpringerLink

Understanding the Unusual Reduction Mechanism of Pd(II) to Pd(I): Uncovering Hidden Species and Implications in Catalytic Cross-Coupling Reactions | Journal of the American Chemical Society
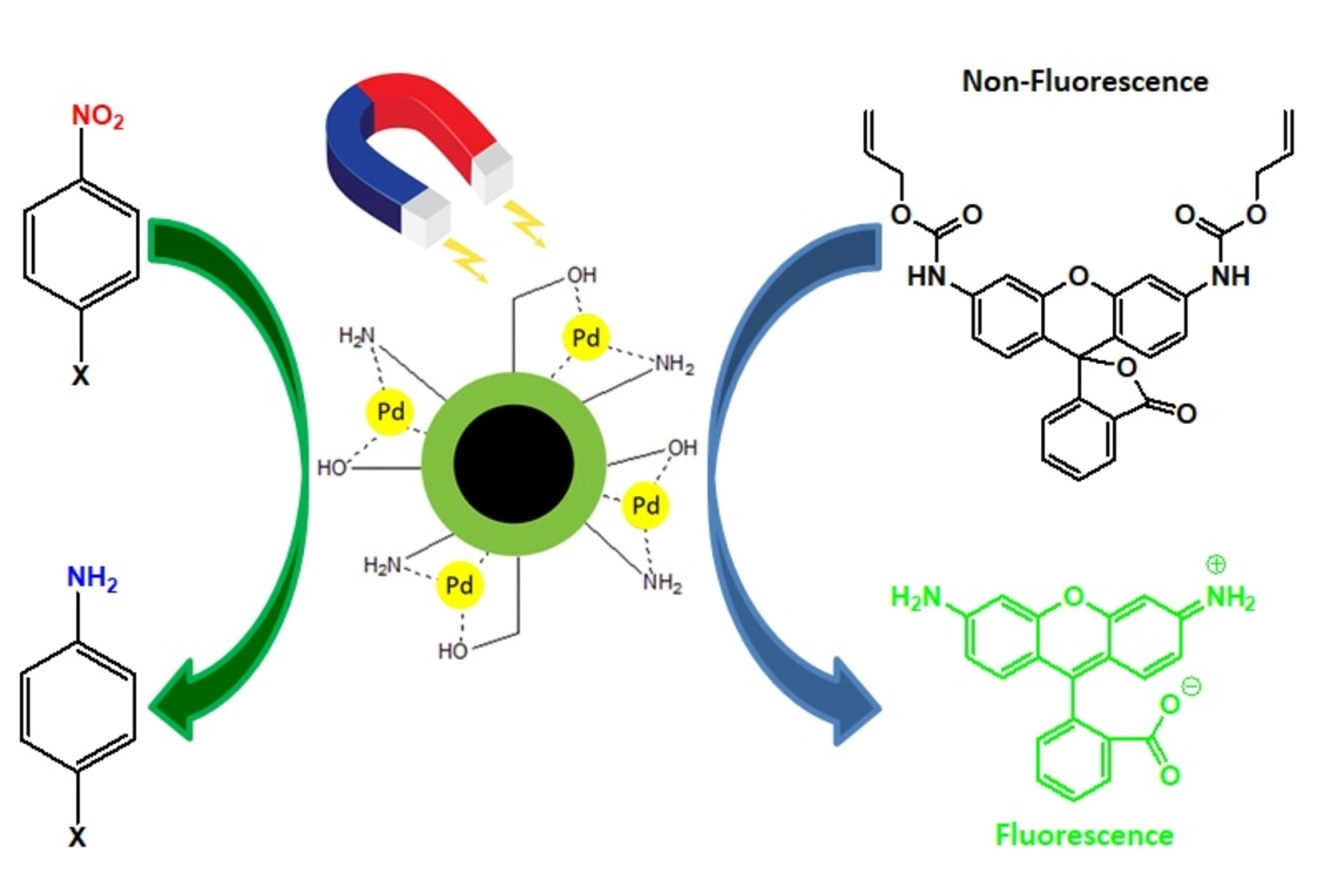
Polymers | Free Full-Text | Palladium Nanoparticles on Chitosan-Coated Superparamagnetic Manganese Ferrite: A Biocompatible Heterogeneous Catalyst for Nitroarene Reduction and Allyl Carbamate Deprotection

Compound A, C_9H_(12), absorbs 3 equivalents of H_2 on catalytic reduction over a palladium catalyst to give B, C_9H_(18). On reaction with KMnO_4, compound A gives, among other things, a ketone that
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img07.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
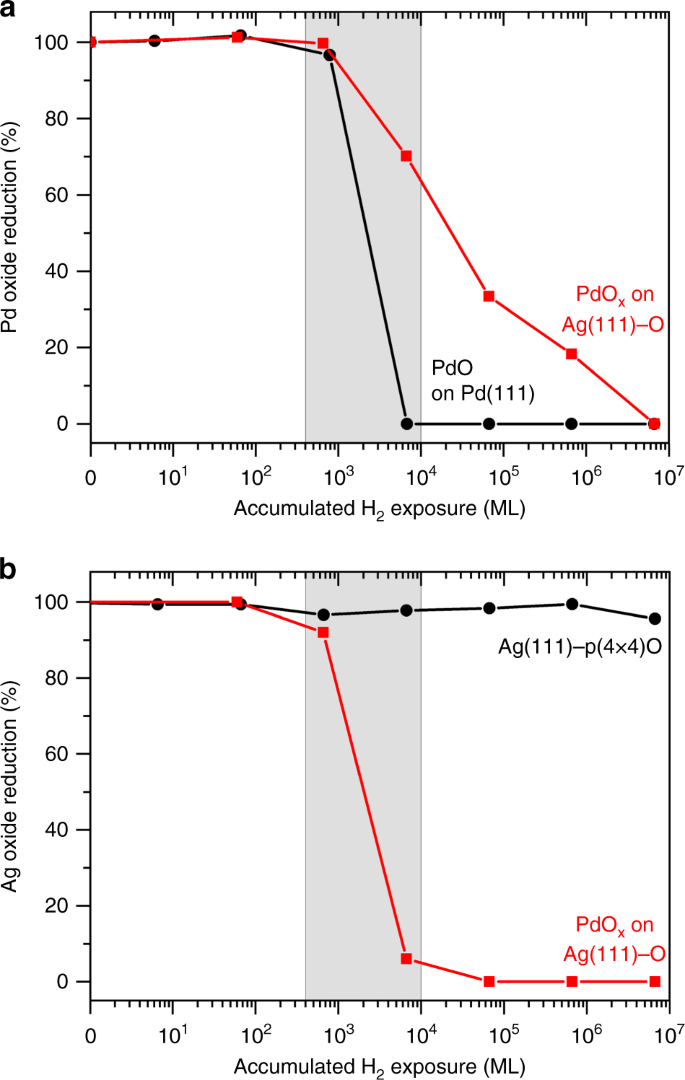
Hydrogen migration at restructuring palladium–silver oxide boundaries dramatically enhances reduction rate of silver oxide | Nature Communications

Electrochemical behavior of CO2 reduction on palladium nanoparticles: Dependence of adsorbed CO on electrode potential - ScienceDirect

Solvent-Induced Reduction of Palladium-Aryls, a Potential Interference in Pd Catalysis | Organometallics

Tandem selective reduction of nitroarenes catalyzed by palladium nanoclusters - Green Chemistry (RSC Publishing)

Solvent-Induced Reduction of Palladium-Aryls, a Potential Interference in Pd Catalysis | Organometallics