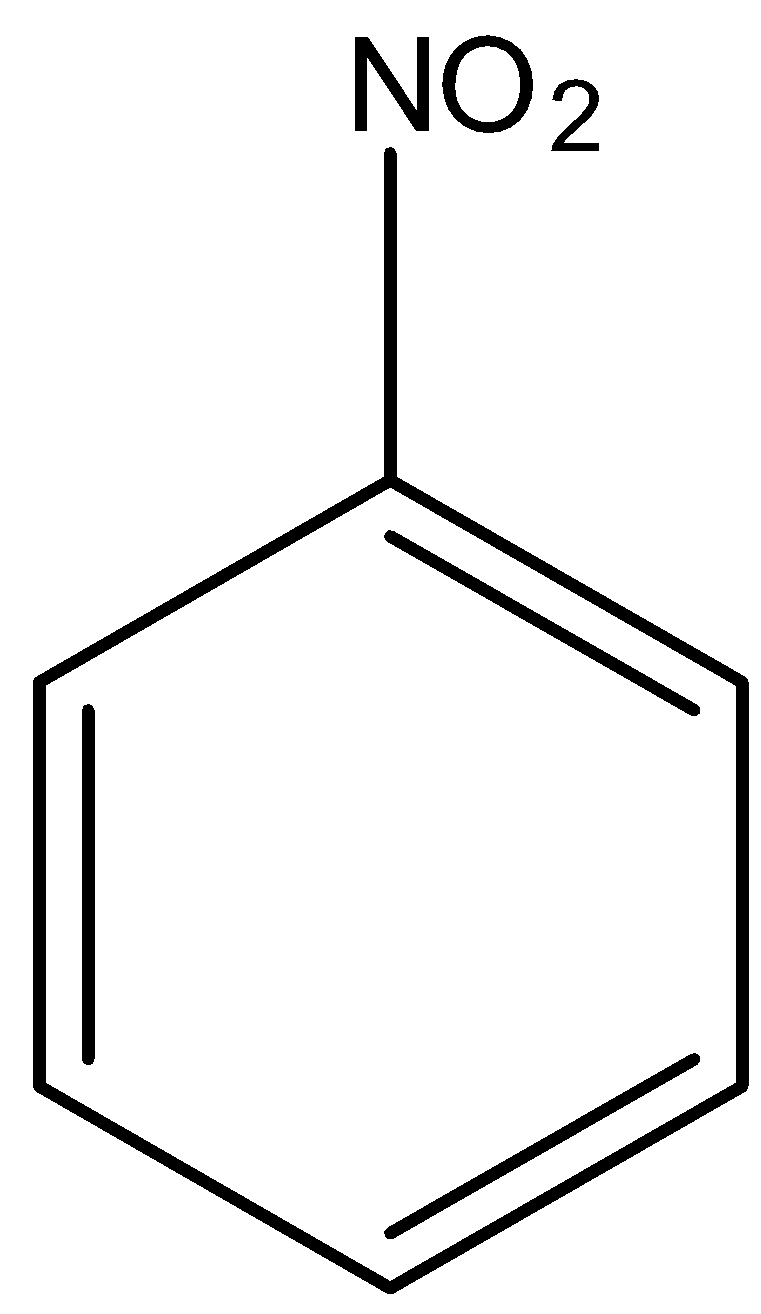
Buff colored precipitate is obtained when $FeC{{l}_{3}}$ is treated with:(a)- \n \n \n \n \n (b)- \n \n \n \n \n (c)- \n \n \n \n \n (d)- \n \n \n \n \
![J Org Chem/Org Lett on Twitter: "The single-component, formally Co(-I) complex Li(THF)3[Co(N2)(PPh3)3] is an efficient catalyst for the coupling of arene and olefin substrates without reactive Grignard reagents or designer ligands. Over J Org Chem/Org Lett on Twitter: "The single-component, formally Co(-I) complex Li(THF)3[Co(N2)(PPh3)3] is an efficient catalyst for the coupling of arene and olefin substrates without reactive Grignard reagents or designer ligands. Over](https://pbs.twimg.com/media/Et4HwT_XcAEBukx.jpg)
J Org Chem/Org Lett on Twitter: "The single-component, formally Co(-I) complex Li(THF)3[Co(N2)(PPh3)3] is an efficient catalyst for the coupling of arene and olefin substrates without reactive Grignard reagents or designer ligands. Over
![J Org Chem/Org Lett on Twitter: "Catechol[4]arene: The missing chiral member of the Calix[4]arene family. A novel, readily available chiral macrocycle. Check it out in #OL: https://t.co/x2xY6Cv64m https://t.co/t9MvnrmpKC" / Twitter J Org Chem/Org Lett on Twitter: "Catechol[4]arene: The missing chiral member of the Calix[4]arene family. A novel, readily available chiral macrocycle. Check it out in #OL: https://t.co/x2xY6Cv64m https://t.co/t9MvnrmpKC" / Twitter](https://pbs.twimg.com/media/EcQhf8VX0AEoBXj.jpg)
J Org Chem/Org Lett on Twitter: "Catechol[4]arene: The missing chiral member of the Calix[4]arene family. A novel, readily available chiral macrocycle. Check it out in #OL: https://t.co/x2xY6Cv64m https://t.co/t9MvnrmpKC" / Twitter

J Org Chem/Org Lett on Twitter: "Multiple electrophilic C–H borylation of arenes using boron triiodide, by Hatakeyama and coworkers in #OL https://t.co/8z53nC4ULB https://t.co/MxolVX1CRf" / Twitter
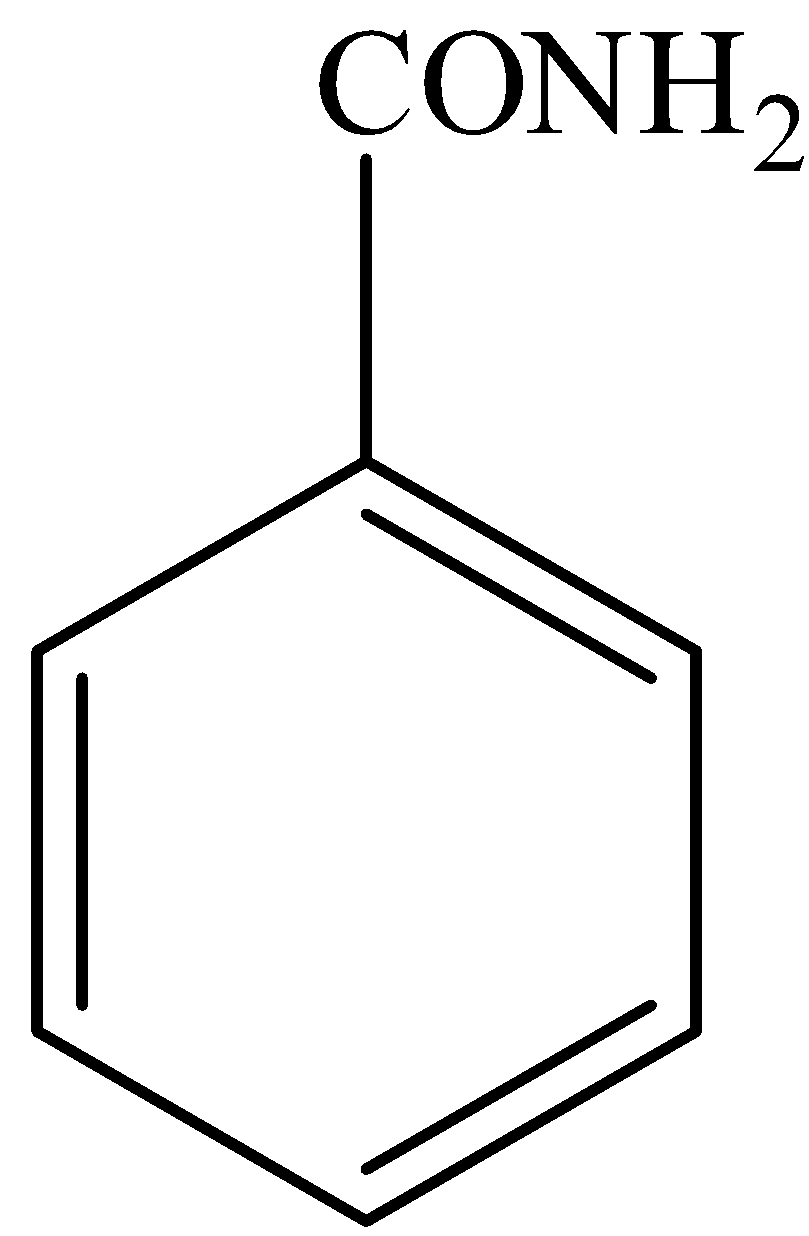
Buff colored precipitate is obtained when $FeC{{l}_{3}}$ is treated with:(a)- \n \n \n \n \n (b)- \n \n \n \n \n (c)- \n \n \n \n \n (d)- \n \n \n \n \
![J Org Chem/Org Lett on Twitter: "A new hybrid macrocycle is born, the Calix[2]naphth[2]arene that can form complexes stabilized by cation···π interactions. Check it out in #OL: https://t.co/2RXxiCchqx https://t.co/dnqQTdYFxM" / Twitter J Org Chem/Org Lett on Twitter: "A new hybrid macrocycle is born, the Calix[2]naphth[2]arene that can form complexes stabilized by cation···π interactions. Check it out in #OL: https://t.co/2RXxiCchqx https://t.co/dnqQTdYFxM" / Twitter](https://pbs.twimg.com/media/Edc83rQXsAE6-K5.jpg)
J Org Chem/Org Lett on Twitter: "A new hybrid macrocycle is born, the Calix[2]naphth[2]arene that can form complexes stabilized by cation···π interactions. Check it out in #OL: https://t.co/2RXxiCchqx https://t.co/dnqQTdYFxM" / Twitter